This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
Finger on the pulse of drug delivery: Preclinical study could pave the way for multiple drug doses in a single injection

Pharmaceutical drugs can save lives, but taking these medications as prescribed—especially among those with chronic conditions—can be challenging, for a variety of different reasons. Improving medication adherence could reduce unfavorable health outcomes, hospitalizations, and preventable deaths, while simultaneously reducing health care costs by up to $300B annually in the United States alone.
One potential way to increase adherence is to reduce how often a person needs to take their medication. This can be achieved through a controlled-release system, where a single injection contains a drug that is continually released in the body over an extended period of time. Unfortunately, many controlled-release systems deliver a substantial portion of their cargo right after injection, which can result in inconsistent drug dosing—more drug is released initially (potentially resulting in toxicity) and less drug is released over time (potentially at too small a dose to be effective). A system that could release discrete doses at specific time points could revolutionize the way drugs are delivered, from multi-dose vaccinations to daily medications.
Responding to this challenge, researchers from Rice University have developed PULSED (for Particles Uniformly Liquified and Sealed to Encapsulate Drugs). Their method, recently reported in Advanced Materials, creates drug-filled microparticles that can be engineered to degrade and release their therapeutic cargo days or weeks after injection. By combining multiple microparticles with different degradation times into a single injection, the researchers could develop a drug formulation that delivers many doses over time.
"As a field, we are continually aiming to develop effective and efficient drug delivery systems that maximize therapeutic benefit and minimize side effects, which can ultimately improve medication adherence," said Luisa Russell, Ph.D., a program director in the Division of Discovery Science & Technology at the National Institute of Biomedical Imaging and Bioengineering (NIBIB). "With several doses combined into a single treatment, the controlled-release system described here could transform the therapeutic landscape, potentially negating the need for frequent drug administration, both at home and at the clinic."
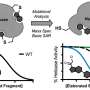
The PULSED microparticles are composed of PLGA, or poly(lactic-co-glycolic acid), a commonly used polymer in a number of FDA-approved devices. PLGA is made up of repeating units of lactic acid and glycolic acid, two molecules that occur naturally in our bodies. By extending the overall length of the polymer, modifying the ratio of lactic acid to glycolic acid, and "capping" the end of the polymer with different molecules, researchers can dictate how long it takes PLGA to disintegrate (and thereby release its therapeutic payload).
"In our case, we can combine populations of microparticles with different PLGA compositions that each release their entire contents at one distinct time point," explained senior study author Kevin McHugh, Ph.D., an assistant professor at Rice University. "This allows us to achieve multiple release events at specific, predetermined times."
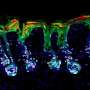
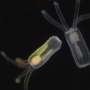
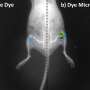
Here's how the drug-loaded microparticles are made: Heated, semiliquid PLGA is pressed into a mold and then cooled, solidifying in the shape of hollow cylinders with an opening at the top. The core of each microparticle is filled with therapeutic cargo, and then the top of the microparticles are heated, causing the PLGA to melt and flow over the opening to seal the drug inside. The initial particles that the researchers developed were 400 micrometers in diameter (for reference, the thickness of a dime is about 1350 micrometers).
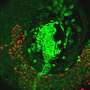
As a first step, the researchers filled four different PLGA microparticle shells with dextran (a type of sugar) that was tagged with a fluorescent molecule, allowing them to easily visualize and measure cargo release. They incubated the microparticles in a body-temperature buffer to mimic real-life conditions and found that the microparticles released their contents at staggered intervals, ranging from roughly eight to 31 days, depending on the PLGA formulation. Importantly, the researchers found that each microparticle formulation released its cargo rapidly, emptying 75% of the dextran over a period of approximately one to three days. They had similar results when they repeated the experiments in mice.
"While we extended the microparticle degradation time to roughly five weeks in this study, we have only begun to tinker with PLGA compositions to further extend their degradation times and subsequent drug release," said McHugh. "Based on our previous work with other systems, we are confident that by changing PLGA length and component ratios, we could extend release to six months and likely much longer."
After the researchers optimized their microparticle fabrication process, they needed to ensure that a pharmaceutical drug was still viable once encased inside. Many drugs—especially biologic agents, which are developed using living components—are sensitive to heat, which the researchers use to seal their microparticles.
What's more, the researchers wanted to be certain that long-term storage inside the human body—conditions that heat and acidify the microparticles—wouldn't adversely affect the drug. They encapsulated bevacizumab (an FDA-approved antibody that is used to treat several types of cancer) into their microparticles along with different kinds of excipients (inactive drug stabilizers) and evaluated the drug's activity. With the right combination of excipients, the microparticles released bioactive and viable bevacizumab, even after weeks under simulated body conditions.
Finally, the researchers wanted to push the envelope and further miniaturize their microparticles. While the original particles could easily flow through an 18-gauge needle, which is routinely used for things like blood collection, smaller needles are preferred for pediatric vaccinations and insulin administration, which typically range from 22 to 31 gauge. Using a smaller mold and the same fabrication method, the researchers were able to shrink the microparticles down to a diameter of 100 micrometers. The loading capacity of these tiny microparticles is 50-fold lower than the original microparticles, but they could potentially be used with even the smallest of commonly used needles, said McHugh. Future work will include evaluating the disintegration time and cargo release of these miniaturized particles.
"Routes of drug administration are often underappreciated in the field of pharmaceutical medicine," McHugh said. "Our study, while still early in development, could reshape how routine vaccinations and recurring medications are delivered, ultimately improving medication adherence and human health."
More information: Tyler P. Graf et al, A Scalable Platform for Fabricating Biodegradable Microparticles with Pulsatile Drug Release, Advanced Materials (2023). DOI: 10.1002/adma.202300228
Journal information: Advanced Materials
Provided by National Institute of Biomedical Imaging and Bioengineering